Esterification Reaction
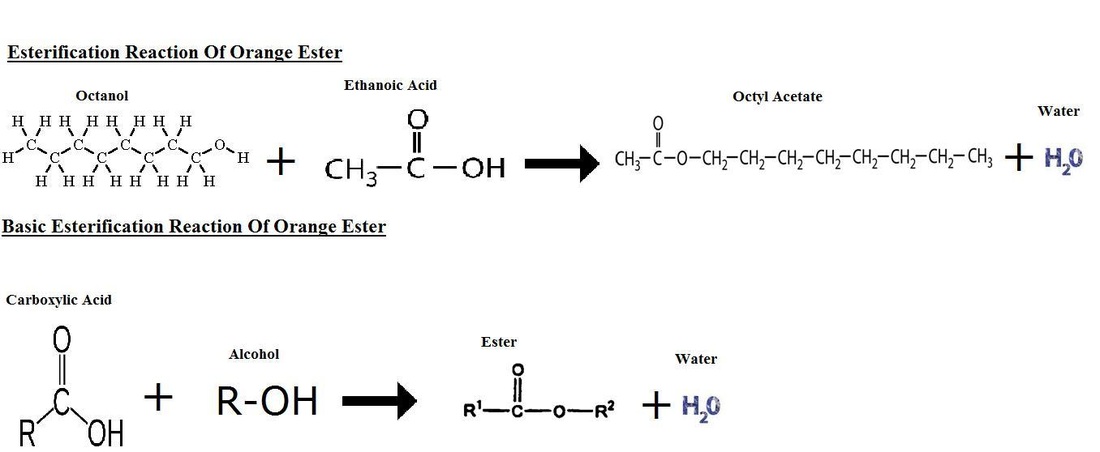
There are two reactions that occurred in this experiment. The first reaction was the esterification reaction where the carboxylic acid combines with an alcohol to form an ester and water. For example, the Ethanoic Acid was combined with the Octanol forms Octyl Acetate (Orange Ester) and water.
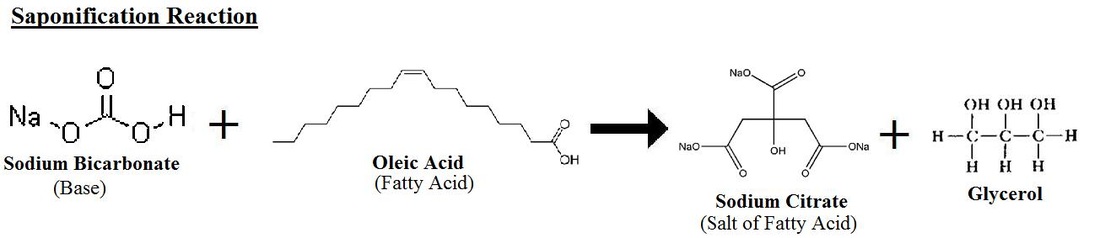
Saponification Reaction
Saponification is a type of esterification. Fats and oils are triglycerides used to combine with a strong base to form salt of the fatty acid (soap) and glycerol. Therefore,a saponification reaction occurs when a fat or oil combines with a base to form a salt and a glycerol. In this case, the Oleic Acid (contained from the olive oil) combined with the sodium bicarbonate to form a glycerol and the sodium citrate salt.
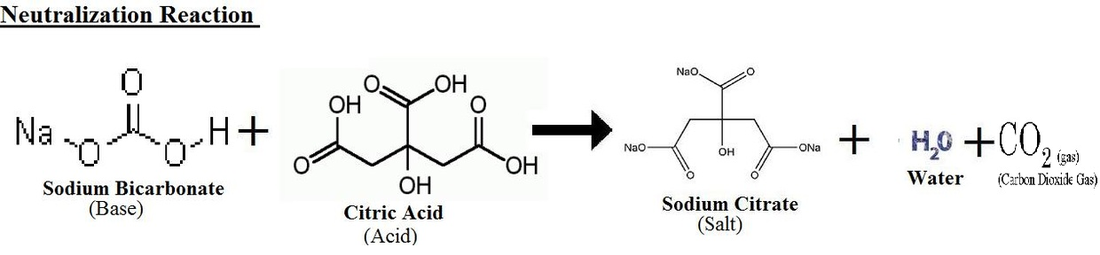
Gas Production Reaction
When the bath bomb in put in water, it starts to fizzle and create bubbles. There is a reaction at occurs with the citric acid and the sodium bicarbonate which produces sodium citrate. This reaction is a neutralization reaction. A neutralization reaction is an acid-base reaction where the sodium bicarbonate is the base reacting with the citric acid. However, the cornstarch, behaves as a liquid absorber to prevent the citric acid and sodium bicarbonate from reacting when liquids like oils are present when creating the bath bomb, before testing to submerge in water. The water is the main ingredient, required for the acid-base reaction to occur liquefies the solid bath bomb and helps to bring the citric acid and sodium bicarbonate ions in motion to collide with each other to produce a reaction. Once the reaction initiates, it allows the production of carbon dioxide gas to occur which appears in the form of bubbles and creates fizziness in the water. Furthermore, the fragrance is also produced which brings a scented smell in the air. Lastly, the oils are produced in the water that moisture the skin.